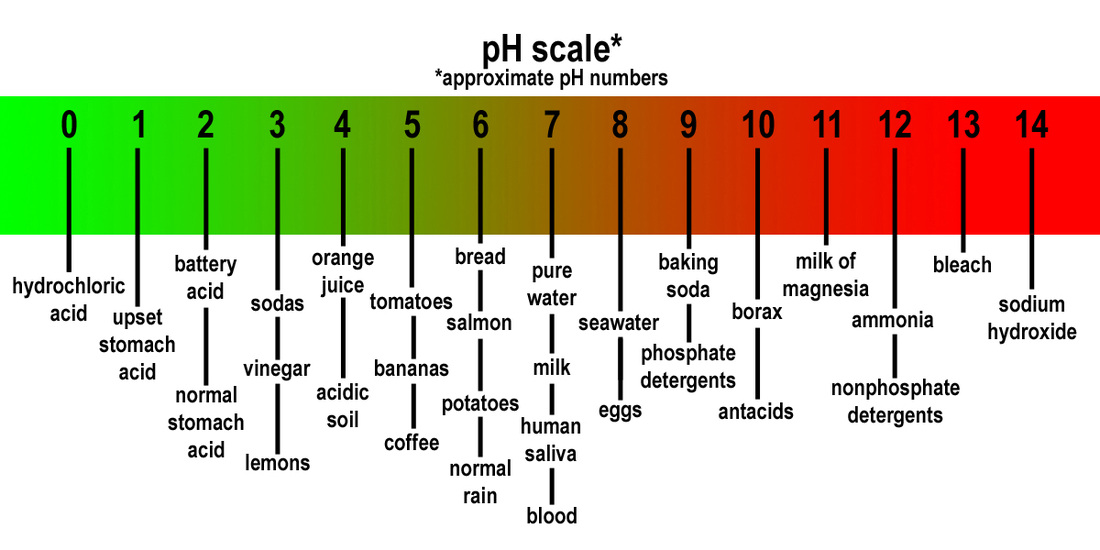
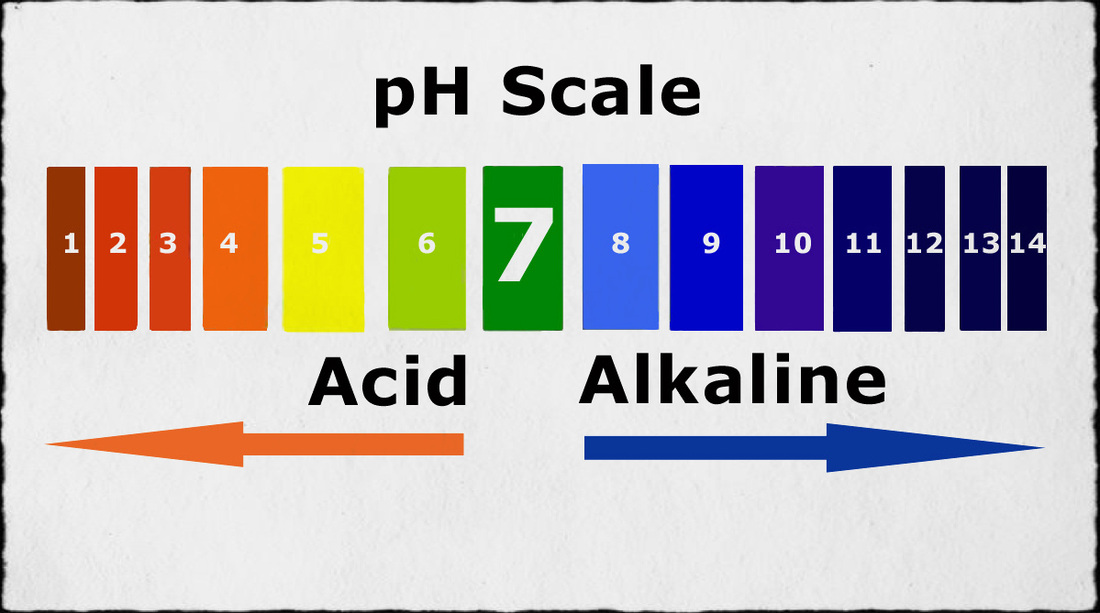
Neutral: A solution that has a pH of 7. It is neither acidic nor basic.
Examples of neutral: Water, Glucose (sugar), and Sodium chloride (table salt).
*The neutralization process happens when an acid and a base interact with each other, causing each of the compounds to lose some of their ions and as a result the remaining solution, it becomes neither an acid nor a base.
A pH indicator is a halochromic chemical compound added in small amounts to a solution so the pH of the solution can be determined visually. An indicator is a large organic molecule that works somewhat like a " color dye". Most of the indicators are themselves weak acids.
Other pH Indicators: Methyl red, 4-Nitrophenol, and Litmus
Examples of neutral: Water, Glucose (sugar), and Sodium chloride (table salt).
*The neutralization process happens when an acid and a base interact with each other, causing each of the compounds to lose some of their ions and as a result the remaining solution, it becomes neither an acid nor a base.
A pH indicator is a halochromic chemical compound added in small amounts to a solution so the pH of the solution can be determined visually. An indicator is a large organic molecule that works somewhat like a " color dye". Most of the indicators are themselves weak acids.
Other pH Indicators: Methyl red, 4-Nitrophenol, and Litmus